Contact Us View Biologics Packages
TA Instruments | Waters Corporation offers a suite of reliable solutions that are essential for ensuring desired product quality and guaranteeing safety and efficacy in the biopharmaceutical industry. Our Microcalorimetry, Thermal Analysis and Rheology solutions correspond to critical quality attributes (CQAs) that must be carefully measured for all kinds of biotherapeutics, including antibodies, cell and gene therapies, and vaccines. Biotherapeutics, unlike chemically synthesized drugs, can be heterogenous in nature and therefore require rigorous quality testing during development and manufacturing.
Characterization of biopharmaceuticals is accomplished with a panel of technologies to understand their basic chemical details:
- Binding affinity, specificity, and stoichiometry (ratio) to the molecular target
- Structure stability, and solvent environment stability.
- Higher Order Structure (HOS)
- Phase transitions
- Viscosity and viscoelastic behavior
- Residual moisture
Explore solutions from TA Instruments | Waters Corporation for biotherapeutics at different development stages. Click on each stage to learn how we can support your next-gen biotherapeutics.

Analytical Solutions for Biotherapeutic Characterization
Understanding cellular pathways is important for addressing key questions in life sciences and combating diseases. Target selection involves identifying a biological target (usually a protein or receptor) that plays a key role in a disease process. Choosing the right target is fundamental because it determines the success of subsequent drug development efforts. The correct target ensures that drugs act specifically on disease-related pathways and off-target effects can be minimized, enhancing drug efficacy and avoiding adverse effects. For instance, a drug targeting a cancer-specific protein is less likely to harm healthy tissues, improving patient safety and overall tolerability. A well-selected target increases the likelihood of therapeutic utility.
Isothermal Titration Calorimetry (ITC) is a powerful technique used to study molecular interactions by measuring the heat exchanged during binding events. By quantifying heat, ITC provides insights into the driving forces behind interactions. Understanding the type and strength of interaction—whether it’s hydrophobic, electrostatic, or other types of interactions—is key. This knowledge enables researchers to design appropriate drugs achieving the desired therapeutic effects by targeting specific points within cellular pathways.
Differential Scanning Calorimetry (DSC) measures the heat absorbed or released during thermal transitions, such as protein unfolding and complements ITC by focusing on the stability and structural characteristics of molecules. Knowing the stability of target molecules is essential for drug development. DSC allows scientists to design better therapeutics and understand differences between native and diseased states. Researchers gain a comprehensive view of molecular interactions by combining ITC and DSC data.
- Binding Affinity
- Thermodynamic Driving Forces
- Stoichiometry
Affinity Range: low-mM, to low-pM
Automation: 96-well plate compatible
- Protein Stability
- Complex Characterization – Binding
- Nucleic Acid Melting
- Phase Transitions
- Heat Capacity
Temperature Range: -10 °C to 130 °C
Automation: 96-well plate compatible
One of the key goals during candidate selection is ensuring effective binding between the drug and the intended target. Binding assays, such as isothermal titration calorimetry (ITC), assess a compound’s affinity for its target. High binding affinity is necessary for in vivo potency and selectivity. ITC uniquely measures the heat of interaction (q) and provides insights into the thermodynamic driving forces by quantifying the contributions from enthalpy (∆H) and entropy (T∆S). Understanding these forces identifies if the binding interaction is specific vs. prone to off-target effects, ensuring that the right candidates advance in development. Other binding assays, such as Surface Plasmon Resonance (SPR), are limited to understanding the binding coefficient despite offering a higher throughput. Relying solely on binding affinity could result in the selection of non-selective candidates, leading to development delays and increased costs.

It becomes fundamental to increase their stability as candidates advance to formulation development. Biological molecules need stabilization for in vitro analysis, extended storage, manufacturing, and processing. In the case of biotherapeutic molecules, unexpected changes in structure could reduce drug product quality and safety. The primary stabilizing forces for biomolecules in solution are the non-covalent interactions between the biomolecule and its immediate solution environment, which means biomolecules rely on excipients such as salts, detergents, sugars, or buffers for stabilization. It is critical to understand how these excipients affect the overall thermal stability and possible function of a therapeutic. Differential scanning calorimetry (DSC) is considered the gold standard for evaluating the thermal stability of biomolecules and works by measuring the heat absorbed or released during thermal transitions, such as protein unfolding. The Nano DSC is designed to characterize the molecular stability of dilute in-solution biomolecules which is particularly valuable when evaluating biomolecule thermal stability, especially at low concentrations.
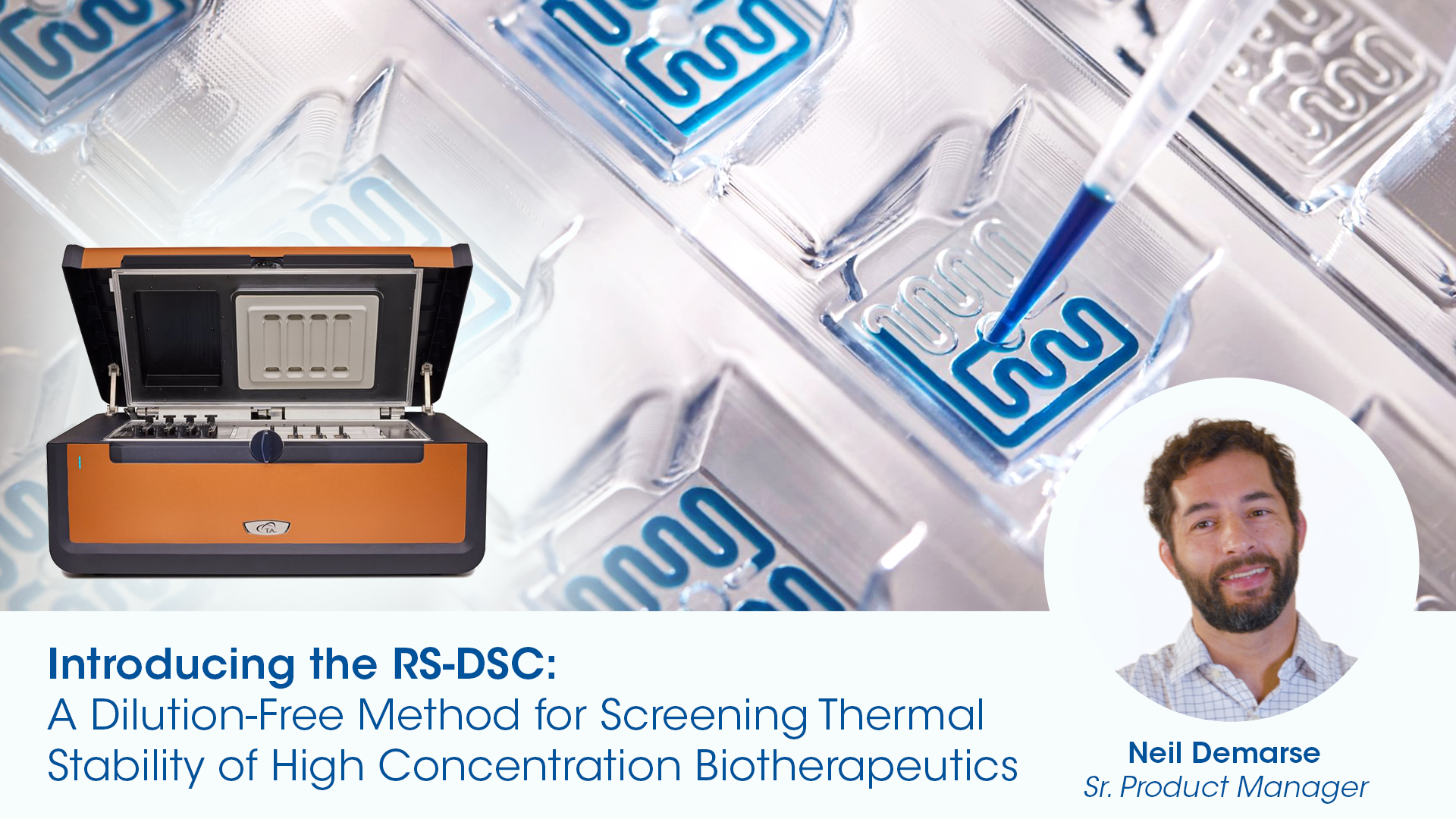
There is a transition to subcutaneous injections for delivery within biologics which necessitates more concentrated formulations. Next, biologic drugs are gradually moving towards subcutaneous injections for delivery, which necessitates more concentrated formulations. Favorable intermolecular interactions occur between proteins and excipients as concentration increases, enhancing molecule stability. Consequently, it becomes crucial to assess thermal stability at dosage concentrations to avoid unexpected issues during development or trials. The Rapid Screening Differential Scanning Calorimeter (RS-DSC) is an ideal method for assessing thermal stability at high dosage concentrations. The RS-DSC eliminates the need for dilution, and its ability to run up to 24 samples simultaneously accelerates the formulation process. Higher concentrations can also lead to reversible or irreversible aggregation, phase separation, precipitation, and increased viscosity. Sample viscosities in the range of 12 cP or lower are preferred for successful subcutaneous delivery. Typically, excipients are added to reduce viscosity during formulation development. The most robust way to measure and optimize viscosity is by utilizing a rheometer to characterize the formulation’s viscosity and viscoelastic behavior.
- Binding Affinity
- Thermodynamic Driving Forces
- Stoichiometry
Affinity Range: low mM – pM
Automation: 96-well plate compatible
- Protein Stability
- Nucleic Acid Melting
- Phase Transitions
- Heat Capacity
Temperature Range: -10 °C to 130 °C
Automation: 96-well plate compatible
Rapid Screening Differential Scanning Calorimeter (RS-DSC)
- Biomolecule Stability
- Tmax
- Enthalpy
Concentration Range: ≥ 20mg/mL – 330+ mg/mL (protein dependent)
Sample Volume: 11 µL
Parallel Measurements: 24
- Viscosity Flow Curve
- Yield Stress
- Viscosity at High Shear Rate
Temperature Range: -150°C to 600°C
Freeze-drying, often called lyophilization, has become a standard process in the pharmaceutical industry for the manufacturing of biologically active substances. Lyophilization offers several benefits including extended stability and simplified storage and shipping but there are limitations due to its high cost in capital and energy, and long processing time. In addition, there are challenges in selecting the process parameters such as time, drying temperature, pressure (vacuum), and component concentration for the lyophilization process. All these parameters must be optimized to: (i) achieve full recovery of activity and complete reconstitution of the often-labile drug, (ii) acceptable appearance of the freeze-dried cake, and (iii) good storage stability.

DSC measurements, such as the glass transition temperature (Tg’), are commonly used to understand characteristics of components like water, bulking agents, buffers and drug, reducing overall costs and achieving high product quality through lyophilization. While low moisture is essential for stability, excessive drying can lead to cake shrinkage; here, complementary thermal analysis techniques like thermogravimetric analysis (TGA) are used to quantify the residual moisture level and optimize process parameters.
Finally, protein solutions are subject to shear stresses during different processes making them susceptible to changes in structure and therefore function. For example, when solutions are being pumped to fill vials or syringes, excessive forces applied during the process could affect the structure of the protein. Other examples of shear forces include shaking and mixing processes. Rheology is a robust way to study the effect of shear flow on proteins. A thorough understanding enables the design of manufacturing processes, conditions, and formulations that optimize yield and stability.
Differential Scanning Calorimeters (DSC)
- Transitions – Tgs, Melting Points, Heat of Fusion
- Lyophilization Optimization
Temperature Range: -180°C to 550°C
Thermogravimetric Analyzers (TGA)
- Residual Moisture (% weight loss)
Temperature Range: 30°C to 1200°C
- Viscosity Flow Curve
- Yield Stress
- Viscosity at High Shear Rate
Temperature Range: -150°C to 600°C
Once a drug formulation passes initial tests, it moves on to manufacturing and quality control evaluation. Manufacturers test critical quality attributes (CQAs) and identify the critical material attributes (CMAs) which must remain stable throughout production and until the drug reaches consumers. Quality control testing detects any batch-to-batch variation, and manufacturing troubleshooting and root cause analysis help manufacturers identify issues in their process. Whether it is ensuring no changes in thermal stability/higher order structure, viscosity of an injectable or the glass transition and residual moisture for a lyophilized product – TA Instruments has a solution for you!

Another key aspect of manufacturing and quality control is compliance with government and industry regulations. By adhering to the guidelines specified in 21 CFR Part 11, companies can guarantee the accuracy and reliability of their electronic records throughout their lifecycle. The optional Guardian package offered in both TRIOS and Nano Software uniquely leverages standard IT security measures and technologies, providing the most cost effective, database-free solution for users who require 21 CFR Part 11 compliance.
- Protein Stability
- Nucleic Acid Melting
- Phase Transitions
- Heat Capacity
Temperature Range: -10 °C to 130 °C
Automation: 96-well plate compatible
- Viscosity Flow Curve
- Yield Stress
- Viscosity at High Shear Rate
Temperature Range: -150°C to 600°C
Differential Scanning Calorimeters (DSC)
- Transitions – Tgs, melting points, heat of fusion
- Lyophilization Optimization
Temperature Range: -180°C to 550°C
Thermogravimetric Analyzers (TGA)
- Residual Moisture (% weight loss)
Temperature Range: 30°C to 1200°C
- Target Selection
-
Understanding cellular pathways is important for addressing key questions in life sciences and combating diseases. Target selection involves identifying a biological target (usually a protein or receptor) that plays a key role in a disease process. Choosing the right target is fundamental because it determines the success of subsequent drug development efforts. The correct target ensures that drugs act specifically on disease-related pathways and off-target effects can be minimized, enhancing drug efficacy and avoiding adverse effects. For instance, a drug targeting a cancer-specific protein is less likely to harm healthy tissues, improving patient safety and overall tolerability. A well-selected target increases the likelihood of therapeutic utility.

Isothermal Titration Calorimetry (ITC) is a powerful technique used to study molecular interactions by measuring the heat exchanged during binding events. By quantifying heat, ITC provides insights into the driving forces behind interactions. Understanding the type and strength of interaction—whether it’s hydrophobic, electrostatic, or other types of interactions—is key. This knowledge enables researchers to design appropriate drugs achieving the desired therapeutic effects by targeting specific points within cellular pathways.
Differential Scanning Calorimetry (DSC) measures the heat absorbed or released during thermal transitions, such as protein unfolding and complements ITC by focusing on the stability and structural characteristics of molecules. Knowing the stability of target molecules is essential for drug development. DSC allows scientists to design better therapeutics and understand differences between native and diseased states. Researchers gain a comprehensive view of molecular interactions by combining ITC and DSC data.
- Binding Affinity
- Thermodynamic Driving Forces
- Stoichiometry
Affinity Range: low-mM, to low-pM
Automation: 96-well plate compatible- Protein Stability
- Complex Characterization – Binding
- Nucleic Acid Melting
- Phase Transitions
- Heat Capacity
Temperature Range: -10 °C to 130 °C
Automation: 96-well plate compatible - Candidate Selection & Formulation Development
-
One of the key goals during candidate selection is ensuring effective binding between the drug and the intended target. Binding assays, such as isothermal titration calorimetry (ITC), assess a compound’s affinity for its target. High binding affinity is necessary for in vivo potency and selectivity. ITC uniquely measures the heat of interaction (q) and provides insights into the thermodynamic driving forces by quantifying the contributions from enthalpy (∆H) and entropy (T∆S). Understanding these forces identifies if the binding interaction is specific vs. prone to off-target effects, ensuring that the right candidates advance in development. Other binding assays, such as Surface Plasmon Resonance (SPR), are limited to understanding the binding coefficient despite offering a higher throughput. Relying solely on binding affinity could result in the selection of non-selective candidates, leading to development delays and increased costs.

It becomes fundamental to increase their stability as candidates advance to formulation development. Biological molecules need stabilization for in vitro analysis, extended storage, manufacturing, and processing. In the case of biotherapeutic molecules, unexpected changes in structure could reduce drug product quality and safety. The primary stabilizing forces for biomolecules in solution are the non-covalent interactions between the biomolecule and its immediate solution environment, which means biomolecules rely on excipients such as salts, detergents, sugars, or buffers for stabilization. It is critical to understand how these excipients affect the overall thermal stability and possible function of a therapeutic. Differential scanning calorimetry (DSC) is considered the gold standard for evaluating the thermal stability of biomolecules and works by measuring the heat absorbed or released during thermal transitions, such as protein unfolding. The Nano DSC is designed to characterize the molecular stability of dilute in-solution biomolecules which is particularly valuable when evaluating biomolecule thermal stability, especially at low concentrations.
There is a transition to subcutaneous injections for delivery within biologics which necessitates more concentrated formulations. Next, biologic drugs are gradually moving towards subcutaneous injections for delivery, which necessitates more concentrated formulations. Favorable intermolecular interactions occur between proteins and excipients as concentration increases, enhancing molecule stability. Consequently, it becomes crucial to assess thermal stability at dosage concentrations to avoid unexpected issues during development or trials. The Rapid Screening Differential Scanning Calorimeter (RS-DSC) is an ideal method for assessing thermal stability at high dosage concentrations. The RS-DSC eliminates the need for dilution, and its ability to run up to 24 samples simultaneously accelerates the formulation process. Higher concentrations can also lead to reversible or irreversible aggregation, phase separation, precipitation, and increased viscosity. Sample viscosities in the range of 12 cP or lower are preferred for successful subcutaneous delivery. Typically, excipients are added to reduce viscosity during formulation development. The most robust way to measure and optimize viscosity is by utilizing a rheometer to characterize the formulation’s viscosity and viscoelastic behavior.
- Binding Affinity
- Thermodynamic Driving Forces
- Stoichiometry
Affinity Range: low mM – pM
Automation: 96-well plate compatible- Protein Stability
- Nucleic Acid Melting
- Phase Transitions
- Heat Capacity
Temperature Range: -10 °C to 130 °C
Automation: 96-well plate compatibleRapid Screening Differential Scanning Calorimeter (RS-DSC)
- Biomolecule Stability
- Tmax
- Enthalpy
Concentration Range: ≥ 20mg/mL – 330+ mg/mL (protein dependent)
Sample Volume: 11 µL
Parallel Measurements: 24- Viscosity Flow Curve
- Yield Stress
- Viscosity at High Shear Rate
Temperature Range: -150°C to 600°C
- Process Development
-
Freeze-drying, often called lyophilization, has become a standard process in the pharmaceutical industry for the manufacturing of biologically active substances. Lyophilization offers several benefits including extended stability and simplified storage and shipping but there are limitations due to its high cost in capital and energy, and long processing time. In addition, there are challenges in selecting the process parameters such as time, drying temperature, pressure (vacuum), and component concentration for the lyophilization process. All these parameters must be optimized to: (i) achieve full recovery of activity and complete reconstitution of the often-labile drug, (ii) acceptable appearance of the freeze-dried cake, and (iii) good storage stability.

DSC measurements, such as the glass transition temperature (Tg’), are commonly used to understand characteristics of components like water, bulking agents, buffers and drug, reducing overall costs and achieving high product quality through lyophilization. While low moisture is essential for stability, excessive drying can lead to cake shrinkage; here, complementary thermal analysis techniques like thermogravimetric analysis (TGA) are used to quantify the residual moisture level and optimize process parameters.
Finally, protein solutions are subject to shear stresses during different processes making them susceptible to changes in structure and therefore function. For example, when solutions are being pumped to fill vials or syringes, excessive forces applied during the process could affect the structure of the protein. Other examples of shear forces include shaking and mixing processes. Rheology is a robust way to study the effect of shear flow on proteins. A thorough understanding enables the design of manufacturing processes, conditions, and formulations that optimize yield and stability.
Differential Scanning Calorimeters (DSC)
- Transitions – Tgs, Melting Points, Heat of Fusion
- Lyophilization Optimization
Temperature Range: -180°C to 550°C
Thermogravimetric Analyzers (TGA)
- Residual Moisture (% weight loss)
Temperature Range: 30°C to 1200°C
- Viscosity Flow Curve
- Yield Stress
- Viscosity at High Shear Rate
Temperature Range: -150°C to 600°C
- Manufacturing & Quality Control
-
Once a drug formulation passes initial tests, it moves on to manufacturing and quality control evaluation. Manufacturers test critical quality attributes (CQAs) and identify the critical material attributes (CMAs) which must remain stable throughout production and until the drug reaches consumers. Quality control testing detects any batch-to-batch variation, and manufacturing troubleshooting and root cause analysis help manufacturers identify issues in their process. Whether it is ensuring no changes in thermal stability/higher order structure, viscosity of an injectable or the glass transition and residual moisture for a lyophilized product – TA Instruments has a solution for you!

Another key aspect of manufacturing and quality control is compliance with government and industry regulations. By adhering to the guidelines specified in 21 CFR Part 11, companies can guarantee the accuracy and reliability of their electronic records throughout their lifecycle. The optional Guardian package offered in both TRIOS and Nano Software uniquely leverages standard IT security measures and technologies, providing the most cost effective, database-free solution for users who require 21 CFR Part 11 compliance.
- Protein Stability
- Nucleic Acid Melting
- Phase Transitions
- Heat Capacity
Temperature Range: -10 °C to 130 °C
Automation: 96-well plate compatible- Viscosity Flow Curve
- Yield Stress
- Viscosity at High Shear Rate
Temperature Range: -150°C to 600°C
Differential Scanning Calorimeters (DSC)
- Transitions – Tgs, melting points, heat of fusion
- Lyophilization Optimization
Temperature Range: -180°C to 550°C
Thermogravimetric Analyzers (TGA)
- Residual Moisture (% weight loss)
Temperature Range: 30°C to 1200°C
Biologics Resources
Biologics Webinars
Biologics Blogs
Biologics Application Notes
- Rapid Thermal Stability Screening of High Concentration Biologic Drugs
- Rapid Thermal Stability Screening and Selection of Monoclonal Antibody Drug Products
- Evaluating AAV Gene Therapy Vectors using Differential Scanning Calorimetry
- Characterizing Virus Structure and Binding
- Characterizing Protein – Protein Interactions by ITC
- Characterization of Biopharmaceutical Stability
- Advances in Native Binding Assays
- Determining Thermal Stability of Antibodies with a Nano DSC
- A Novel Thermodynamic Assay for Predicting and Monitoring Biomolecular Structure Stability
- Workflow Solutions: Choosing the Right ITC Binding Experiment
- Nano DSC: What to Consider when Choosing a Baseline and Model
- Isothermal Titration Calorimetry in Organic Solvent Systems
- View All Application Notes