The TAM IV- 48 expands the TAM IV capabilities as a high-throughput microcalorimeter system that can be configured to accommodate up to 48 individual calorimeters.
Sales Promotions
TAM IV-48 is a high-throughput version of the TAM IV and can accommodate up to 48 individual 4 mL Minicalorimeters.
The large number of independent and parallel sample-measurements makes this system the optimal solution for sample screening, formulation, and process development. It is equally useful in research laboratories as in industrial laboratories including pharmaceutical production control.
Features & Benefits
Features & Benefits
- New and more responsive temperature control provides rapid thermal equilibration and extended temperature range of 4 °C to 150 °C for real-world cold storage applications.
- Unmatched temperature stability for accurate heat flow measurement in experiments that last anywhere from a few hours to days and weeks
- 48 calorimeter positions that can be equipped with up to 48 4 mL Minicalorimeters, each of which operates simultaneously and independently for high-throughput microcalorimetric measurements
Calorimeters

From one and up to 48 4 mL Minicalorimeters can be installed in the TAM IV-48 thermostat.
A Minicalorimeter is a 4 mL calorimeter with a special design to reduce the space occupied by the calorimeter inside the TAM thermostat. The permanently mounted reference is positioned below the sample and is optimized to match the most typical sample. This space-saving over-under design make it possible for high sample throughput by maximizing the number of calorimeters in the thermostat. Each calorimeter can be used independent, the only thing they have in common is the measuring temperature.
Minicalorimeter
 Minicalorimeter
Minicalorimeter
The Minicalorimeter is a 4 ml Microcalorimeter with a special design to reduce the space occupied by the calorimeter inside the thermostat. The reference is positioned below the sample ampoule which allows up to 48 Minicalorimeters to be positioned in the thermostat. The Minicalorimeter is used in the Multicalorimeter and in the TAM 48 thermostat. It has been designed for increased sample throughput and is recommended for compatibility and stability testing.
TAM Ampoules
TAM Ampoules are used to contain the sample in the calorimeter during measurement. The ampoules are of two basic types; Closed and Open. In the closed ampoules, no manipulation to the sample is done during the measurement. In the open ampoules, also referred to as the Micro Reaction System, the sample can be manipulated after insertion into the calorimeter. The most common ampoule types for the TAM IV-48 are the closed ampoules
Closed Ampoules
Closed Ampoules
Disposable Crimp Seal Glass Ampoules
Disposable ampoules are the most convenient to use since they can be thrown away after use and no cleaning is required. The crimp seal ampoule is perfect for experiments at lower temperature ranges. They are available in 3 and 4 mL sizes.
Stainless Steel Ampoules
Available in regular stainless steel or Hastelloy, these ampoules are used for samples that either react with glass or are to be investigated at high temperatures, Ampoule lids are screw top and are available in 4 mL sizes.
Heat Sealed Ampoules
The heat seal ampoules are sealed by melting the glass at the top of the ampoule. These are recommended when rubber caps would be affected by gases or liquid involved in a reaction. The reactants are completely surrounded by glass. They are available for a maximum sample volume of 5 ml.
The versatility of the TAM system provides for a common platform through which to perform a wide variety of calorimetric experiments.
Isothermal Calorimetry
ISOTHERMAL CALORIMETRY
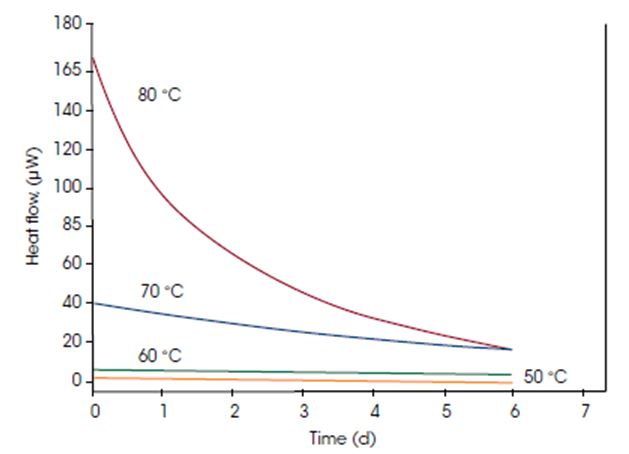
The most common experimental mode for microcalorimetry is the isothermal mode, where the sample is held at a constant temperature while the heat produced or consumed by the sample is measured. Both short- and long-term measurements (hours to weeks) can be performed with exceptional stability and reliability. The example shows how the reaction rate increases with increased temperature.
Scanning Calorimetry
SCANNING CALORIMETRY
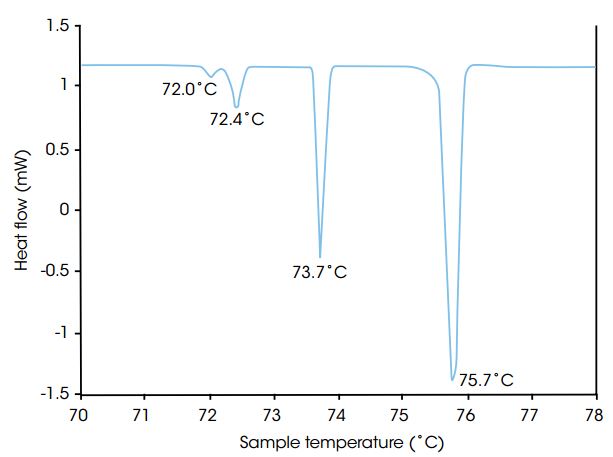
The sample is ramped slowly up and/or down in temperature while the heat produced or consumed by the sample is measured. Experiments performed under extremely slow (maximum 2 °C/h) and well controlled scanning conditions provides greater resolution of phase transitions compared to conventional DSC instruments. The slow scan rate allows large samples to be analyzed close to thermal, physical and chemical equilibrium
The example show a slow scanning experiment resolving four endothermic transitions of n-hexatriacontane
- Description
-
TAM IV-48 is a high-throughput version of the TAM IV and can accommodate up to 48 individual 4 mL Minicalorimeters.
The large number of independent and parallel sample-measurements makes this system the optimal solution for sample screening, formulation, and process development. It is equally useful in research laboratories as in industrial laboratories including pharmaceutical production control.
- Features
-
Features & Benefits
Features & Benefits
- New and more responsive temperature control provides rapid thermal equilibration and extended temperature range of 4 °C to 150 °C for real-world cold storage applications.
- Unmatched temperature stability for accurate heat flow measurement in experiments that last anywhere from a few hours to days and weeks
- 48 calorimeter positions that can be equipped with up to 48 4 mL Minicalorimeters, each of which operates simultaneously and independently for high-throughput microcalorimetric measurements
- Calorimeters
-
Calorimeters

From one and up to 48 4 mL Minicalorimeters can be installed in the TAM IV-48 thermostat.
A Minicalorimeter is a 4 mL calorimeter with a special design to reduce the space occupied by the calorimeter inside the TAM thermostat. The permanently mounted reference is positioned below the sample and is optimized to match the most typical sample. This space-saving over-under design make it possible for high sample throughput by maximizing the number of calorimeters in the thermostat. Each calorimeter can be used independent, the only thing they have in common is the measuring temperature.
Minicalorimeter
 Minicalorimeter
MinicalorimeterThe Minicalorimeter is a 4 ml Microcalorimeter with a special design to reduce the space occupied by the calorimeter inside the thermostat. The reference is positioned below the sample ampoule which allows up to 48 Minicalorimeters to be positioned in the thermostat. The Minicalorimeter is used in the Multicalorimeter and in the TAM 48 thermostat. It has been designed for increased sample throughput and is recommended for compatibility and stability testing.
- Ampoules
-
TAM Ampoules
TAM Ampoules are used to contain the sample in the calorimeter during measurement. The ampoules are of two basic types; Closed and Open. In the closed ampoules, no manipulation to the sample is done during the measurement. In the open ampoules, also referred to as the Micro Reaction System, the sample can be manipulated after insertion into the calorimeter. The most common ampoule types for the TAM IV-48 are the closed ampoules
Closed Ampoules
Closed Ampoules
Disposable Crimp Seal Glass Ampoules
Disposable ampoules are the most convenient to use since they can be thrown away after use and no cleaning is required. The crimp seal ampoule is perfect for experiments at lower temperature ranges. They are available in 3 and 4 mL sizes.
Stainless Steel Ampoules
Available in regular stainless steel or Hastelloy, these ampoules are used for samples that either react with glass or are to be investigated at high temperatures, Ampoule lids are screw top and are available in 4 mL sizes.
Heat Sealed Ampoules
The heat seal ampoules are sealed by melting the glass at the top of the ampoule. These are recommended when rubber caps would be affected by gases or liquid involved in a reaction. The reactants are completely surrounded by glass. They are available for a maximum sample volume of 5 ml.
- Experiments
-
The versatility of the TAM system provides for a common platform through which to perform a wide variety of calorimetric experiments.
Isothermal Calorimetry
ISOTHERMAL CALORIMETRY

The most common experimental mode for microcalorimetry is the isothermal mode, where the sample is held at a constant temperature while the heat produced or consumed by the sample is measured. Both short- and long-term measurements (hours to weeks) can be performed with exceptional stability and reliability. The example shows how the reaction rate increases with increased temperature.
Scanning Calorimetry
SCANNING CALORIMETRY

The sample is ramped slowly up and/or down in temperature while the heat produced or consumed by the sample is measured. Experiments performed under extremely slow (maximum 2 °C/h) and well controlled scanning conditions provides greater resolution of phase transitions compared to conventional DSC instruments. The slow scan rate allows large samples to be analyzed close to thermal, physical and chemical equilibrium
The example show a slow scanning experiment resolving four endothermic transitions of n-hexatriacontane
- Resources











