Keywords: thermal analysis, sorption analysis, sorption analyzers, moisture, GAB, protein
TA363
Abstract
Adsorption/desorption isotherm data collected from moisture sorption instruments such as the Q5000SA or the VTI-SA can be theoretically modeled to calculate properties such as surface area or heat of adsorption. This paper illustrates a GAB analysis of the adsorption of water on a protein sample.
Introduction
In 1938 Brunauer, Emmett and Teller demonstrated, with the help of a few simplifying assumptions, that it was possible to carry out an isotherm derivation for multi-molecular adsorption layers.1 The resultant BET method has been used mainly for determining the surface area of solids from the nitrogen adsorption isotherm at liquid nitrogen temperatures but the method has been used with other gases and vapors for the same purpose.
The equation in its linear form can be written as:
P/Po/W(1-P/Po) = 1/WmC +((C-1)/WmC)P/Po (1)
Where P/Po is the relative pressure of the adsorbate gas or vapor, W is the mass of the adsorbed gas or vapor per unit mass of adsorbent, Wm is the monolayer capacity or mass of adsorbate needed to form a monolayer on the sample’s surface and C is the so-called BET constant. This constant is directly proportional to the heat of adsorption and therefore a measure of the strength of the interaction between the surface and the adsorbed gas or vapor.
Because of the nature of the BET equation, correlation is very good up to 50% RH values. Beyond 50% RH the BET equation typically overestimates the adsorbate uptake. This is due to its failure to take into account the change in adsorption dynamics once a monolayer of adsorbate has formed.
These shortcomings of the BET equation were recognized soon after the model was first published. Of the several modifications proposed, the one suggested by Guggenheim, Anderson and deBoer has been the most successful. This model (the GAB equation) has been used extensively in the food and pharmaceutical industries to study water sorption isotherms on solids. In areas outside these industries, like catalysis, it is virtually unknown.
The GAB equation differs from the BET analog in the introduction of one constant, k, which is intended to correct for the sorption state of the sorbed molecules beyond the first layer, so their character lies somewhere between the strongly bound first layer and the totally free liquid. By introducing this constant, the range of applicability of the original BET equation is extended to values above 80%RH or P/Po above 0.8
The GAB equation is written in the following manner:
W = (WmCkP/Po)/(1-k(P/Po)(1-kP/Po + CkP/Po)) (2)
where Wm and C are analogous to the BET equation. The value of k is always less than 1 (for k=1, one obtains the BET equation) and takes characteristic values for certain materials. The equation can be linearized like the BET equation to solve for the three constants but it is easier to use a non-linear, multiple variable regression method.
Results and Discussion
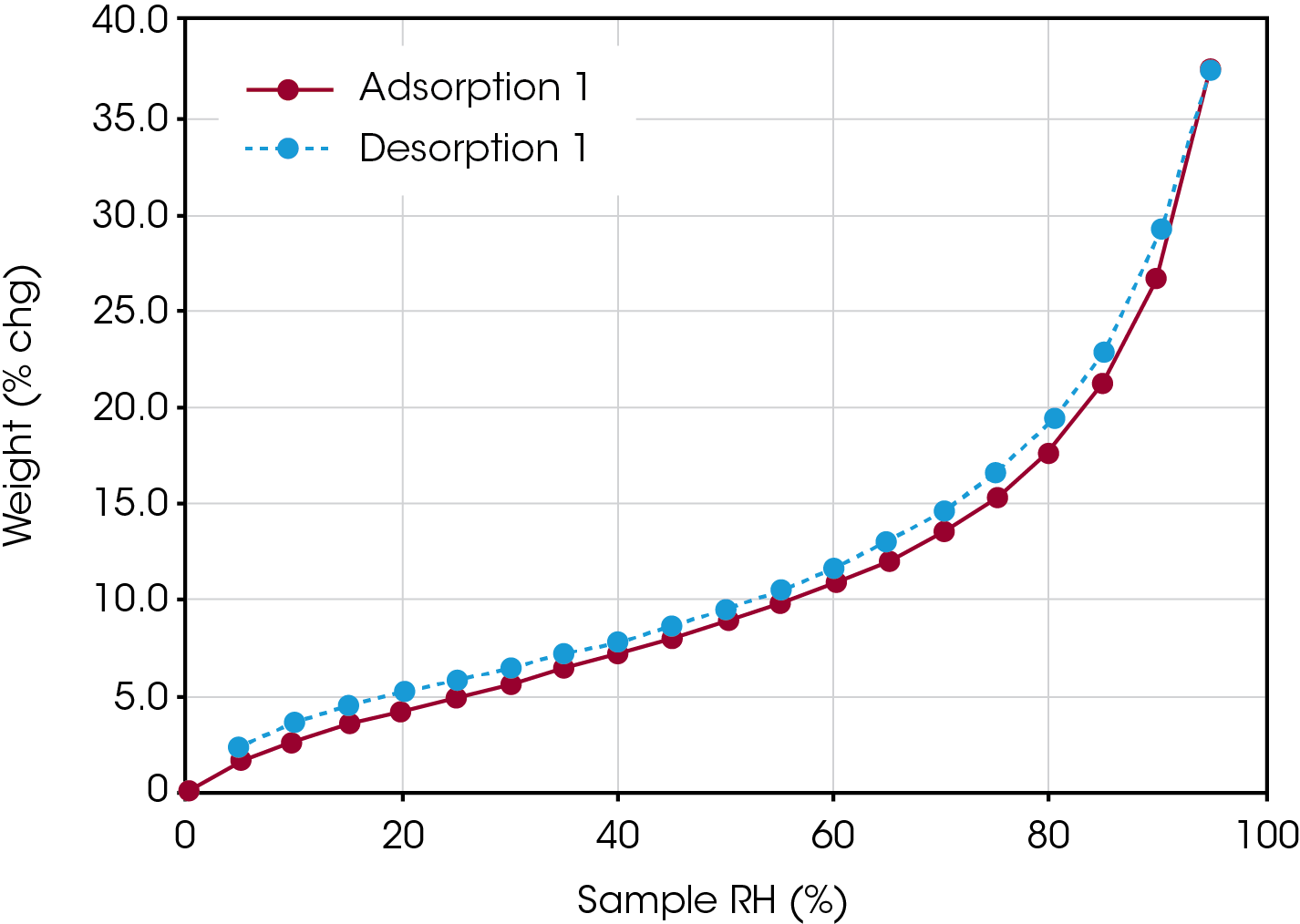
Figure 1 contains an adsorption/desorption isotherm of water on a protein sample collected at 25°C. A classic Type II isotherm is displayed, with less than 15% mass change up to a relative humidity of 70% and then more than 15% mass change from 70-95% relative humidity.
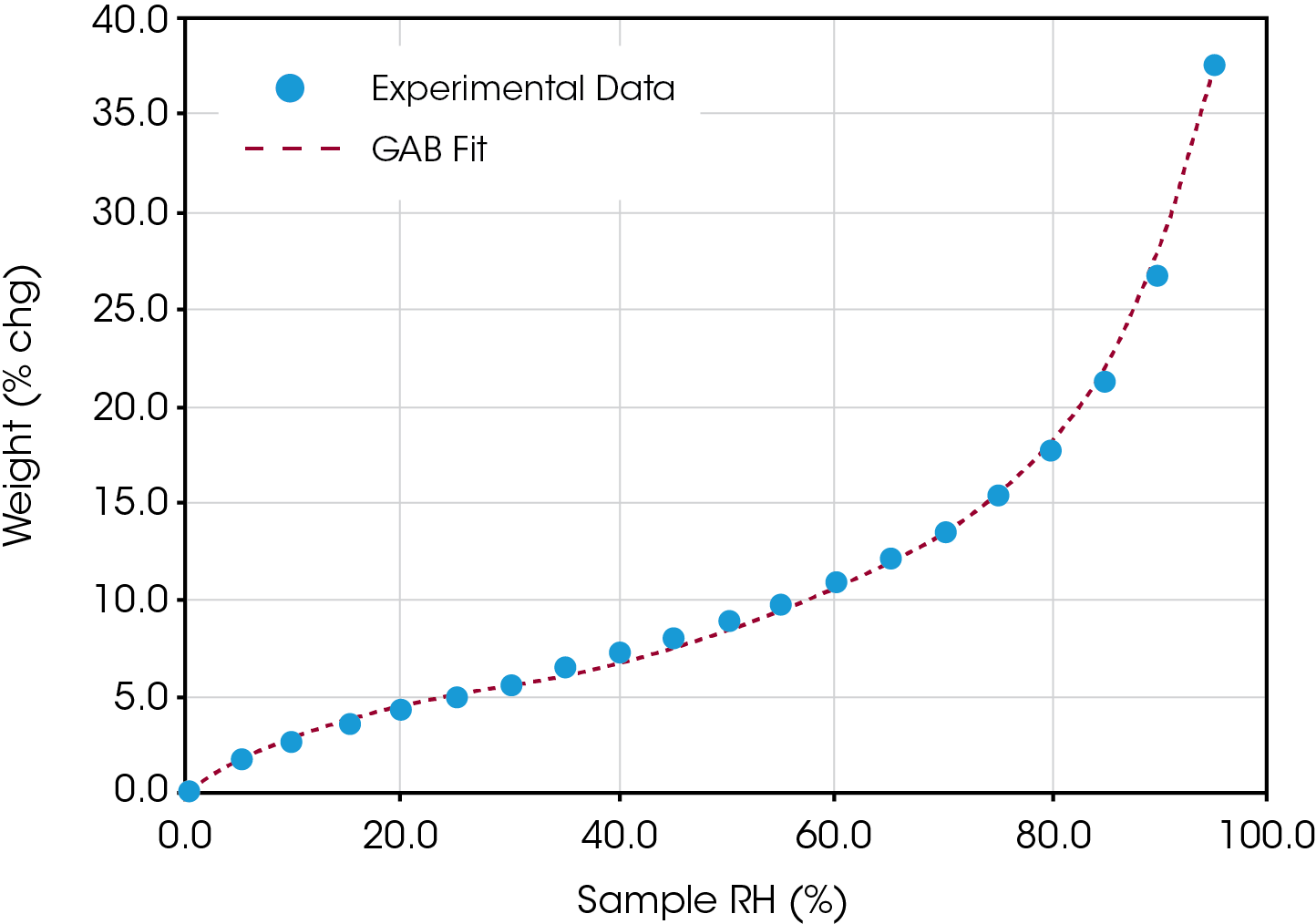
Figure 2 on the next page shows the theoretical GAB fit overlaid with the experimental data of % weight change versus relative humidity. The fit is excellent over the entire range of the experiment – up to 95% humidity. The fit also produces the following results:
Wm = 0.00281 moles/g
C = 11.115
k = 0.913
From the monolayer capacity, Wm, the surface area (SA) is easily calculated by the following formula:
SA = Wm • N • Am
Where SA is the surface area of the solid, in m2/g, Wm is the monolayer capacity, in moles/g, N is Avogadro’s number (6.0221×1023 molecules/mole) and Am the cross sectional area of the adsorbate molecule, in this case 12.5×10-20 m2/molecule for water. Thus:
SA = (0.00281)(6.0221×1023 )(12.5×10-20 ) = 211.5 m2/g
References
- Brunauer, Emmet and Teller, J.Am.Chem.Soc., 60, pp. 686, 1938
Click here to download the printable version of this application note.

