Keywords: Melting Temperature, Heat of Fusion, Apparent Melting, Thermodynamic Melting, Kinetic Process, Differential Scanning Calorimetry, Decomposition, Drug Excipient Incompatibility, Thermogravimetric Analysis, Modulated DSC (MDSC®), Quasi-isothermal MDSC®
TA404
Abstract
Drug-excipient incompatibility is a term used to describe chemical interaction between an active pharmaceutical ingredient (API) and another material (excipient) that is added to perform a specific function in the final dosage form. These functions can include lubrication during manufacturing, thermal or oxidative stability, binder, color, sweetness, bioavailability etc. The excipient is typically identified as an inert ingredient, and is expected to not affect the chemistry or physical properties of the API. Verifying that the excipient is not affecting the API typically requires months of testing and significant use of resources such as manpower, oven usage and analytical time.
The proposed method uses the approach of apparent melting (see TA401) and provides a rapid screening protocol that can quickly identify problem excipients and reduce detection of drug-excipient incompatibility to a few days.
This method does not eliminate the need to do long term oven aging and analytical testing of a final candidate dosage form but should improve the probability of a successful development.
Introduction
Thermal analysis techniques such as Differential Scanning Calorimetry (DSC), Modulated DSC (MDSC®) and Thermogravimetric Analysis (TGA) have been used for almost fifty years to characterize the structure and thermal properties of materials used by the pharmaceutical industry. However, until now, attempts to develop an approach to detect drug-excipient incompatibility have been largely unsuccessful. Reasons for this failure are beyond the scope of this paper but one of the primary causes was a lack of understanding of how kinetic processes such as chemical interaction affect crystallinity and the heat flow signal observed in DSC experiments.
Recent work by researchers at the University of Illinois, Urbana-Champaign1,2,3 has shown that “apparent melting” occurs when chemical processes such as decomposition occur in crystalline structure. The apparent melting peak occurs below the thermodynamic melting of these materials and is time dependent, i.e. shifts to higher temperature at higher heating rates. These endothermic peaks look just like an expected melting peak and are often misinterpreted as thermodynamic melting. Their work also showed how the rate of crystallinity loss due to apparent melting could be measured as a function of time at specific temperatures using the approach of quasi-isothermal Modulated DSC.
The purpose of this paper is to explain and illustrate a systematic thermal analysis approach (DSC, MDSC®, and TGA) for detecting drug-excipient incompatibility and then optionally exploring the rate of reaction (kinetics) and reaction mechanism (chemistry). This approach can be summarized as follows:
- TGA analysis at 10°C/min on API and excipient
- DSC analysis at multiple heating rates on API and excipient
- DSC analysis at 1°C/min on a 50/50 (wt.) mixture of API and excipient. If apparent melting is detected (temperature shift of endothermic peak) then optional testing can be performed
- Optional analyses to measure reaction kinetics and/or identify reaction mechanism
- Use of quasi-isothermal MDSC at three temperatures to create an Arrhenius plot that can be used to obtain reaction kinetics (activation energy and pre-exponential factor)
- Use of quasi-isothermal MDSC at a single temperature to create a plot of heat capacity vs. time that can be used to calculate percent conversion vs. time at that temperature. That plot can be used to prepare samples of different percent conversion in an oven so that they can be used with any analytical technique (HPLC, IR, MS, XRD etc.) to investigate the reaction mechanism
Details
Step 1: TGA Analysis at 10°C/min on API and Excipient
The purpose of doing TGA is to measure and verify composition, and to determine the temperature of bulk mass loss due to decomposition. Specific analyses include:
- Free/unbound water. This is water that could plasticize amorphous material or cause hydrolysis in some component. Plasticization of amorphous structure lowers the glass transition temperature and increases molecular mobility.
- Presence of hydrate forms. Hydrates typically undergo dehydration at temperatures below the melting point of anhydrous structure. This dehydration causes a creation of free/unbound water and results in formation of amorphous structure which is less stable than crystalline structure.
- Temperature of bulk mass loss. This is an important determination because it is necessary to do the DSC experiments in hermetic pans. These pans will begin to leak once the internal pressure exceeds approximately 50psi. Leaking of decomposition products into the DSC is a problem since it may affect the baseline performance of future experiments and cause contamination. With 5mg samples, it is recommended to limit the DSC upper temperature to the temperature where no more than 10% weight loss occurs in the TGA experiment.
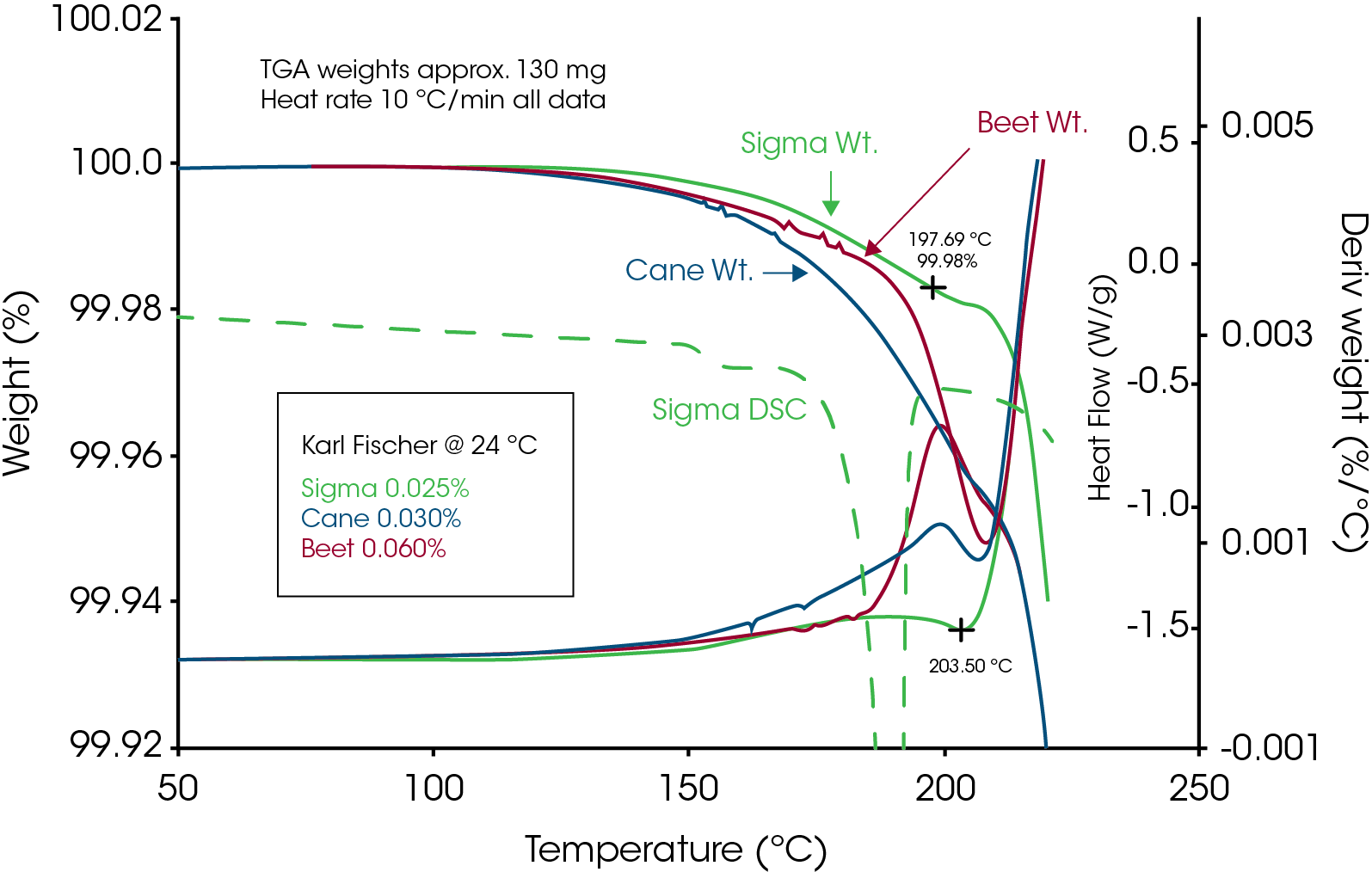
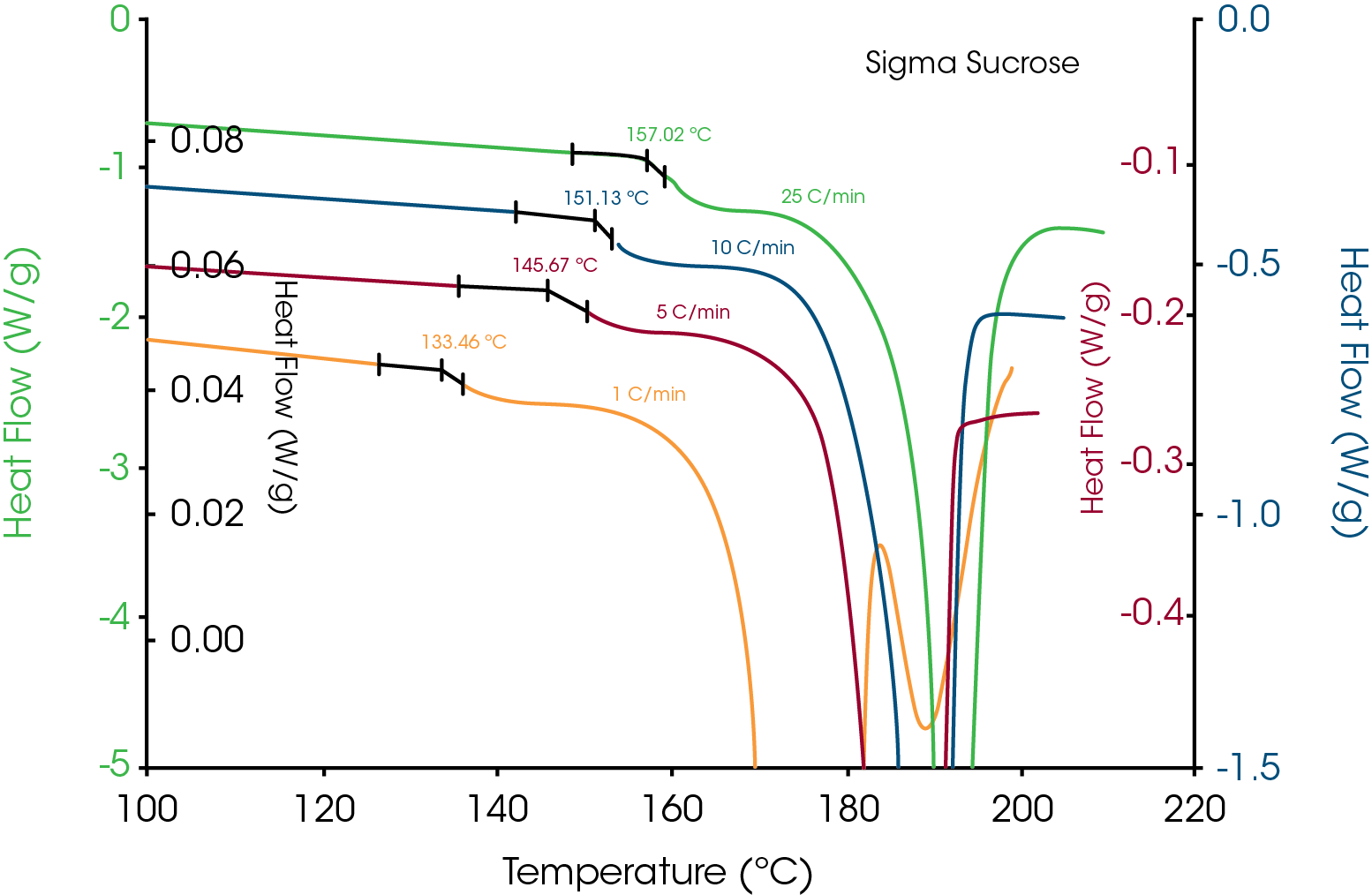
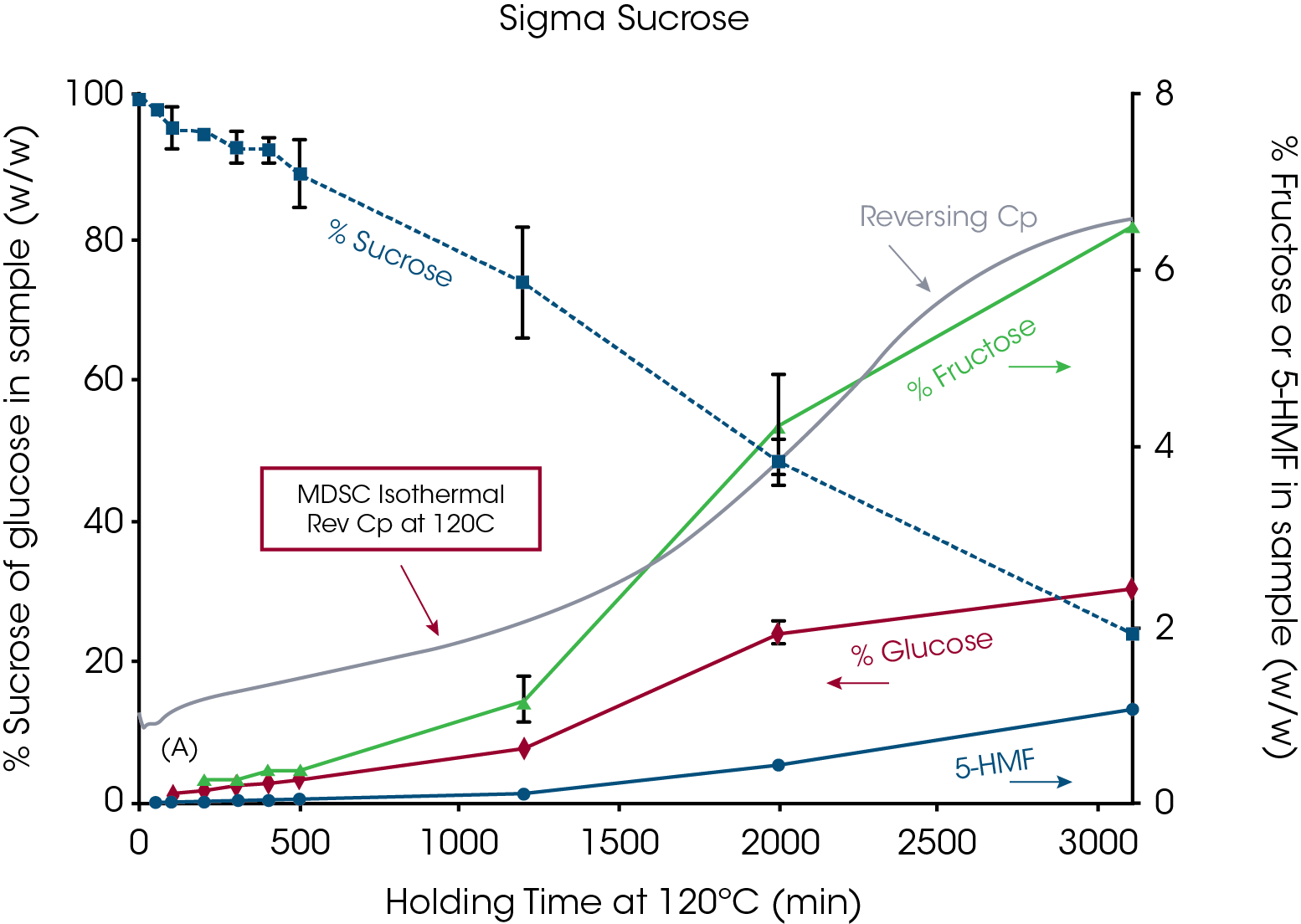
Before illustrating some of the above measurements, it is important to address a common misunderstanding of the ability of TGA to reliably measure decomposition temperature. This misunderstanding is based on the belief that decomposition always causes weight loss which is just not true. Weight loss only occurs if the decomposition mechanism results in formation of volatile decomposition products. This was shown by Lee et.al.1for sucrose. Figure 1 shows TGA data for sucrose obtained from three sources, sugar cane, sugar beet and analytical grade. That data was created with sample weights of approximately 130mg so that TGA moisture analysis could be compared with data from Karl Fisher titration. The maximum weight loss in the three TGA experiments was less than 0.04% at 200°C which agreed with the Karl Fisher results. Based on the TGA derivative signals, significant weight loss does not begin until about 200°C. For comparison, the DSC data overlaid on Figure 1 (also at 10°C/min) shows that the endothermic peak associated with loss of crystalline structure is completed by that temperature. This kind of data convinced many researchers in the past that the melting of sucrose must be thermodynamic because loss of crystalline structure occurred prior to decomposition. The work by Lee et.al.1 showed that sucrose undergoes apparent melting as seen in Figure 2 and that loss of crystalline structure due to decomposition occurs as low as 120°C during isothermal experiments (Figure 14).
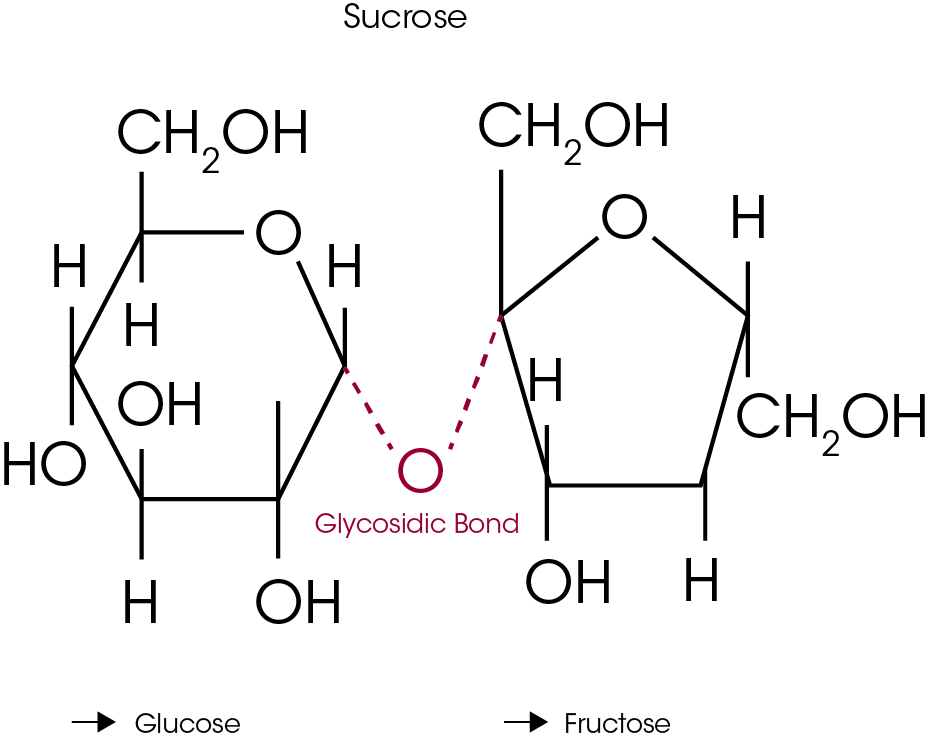
Results that show decomposition begins as low as 120°C by DSC but weight loss due to TGA decomposition begins near 200°C seem to be in conflict. This apparent conflict is the result of believing that decomposition always causes weight loss. As will be discussed and illustrated later in this paper, both chemical and kinetic analyses confirm that decomposition does occur at temperatures as low as 120°C. Understanding the decomposition mechanism resolves the issue because it shows that decomposition creates fructose and glucose which are not volatile. The mechanism of decomposition is cleavage of the glycosidic bond as shown in Figure 3.
Although it is not recommended to identify decomposition using DSC, it is the most reliable technique for detecting decomposition and chemical interaction in crystalline materials since those processes result in a loss of crystalline structure and a change in chemistry.
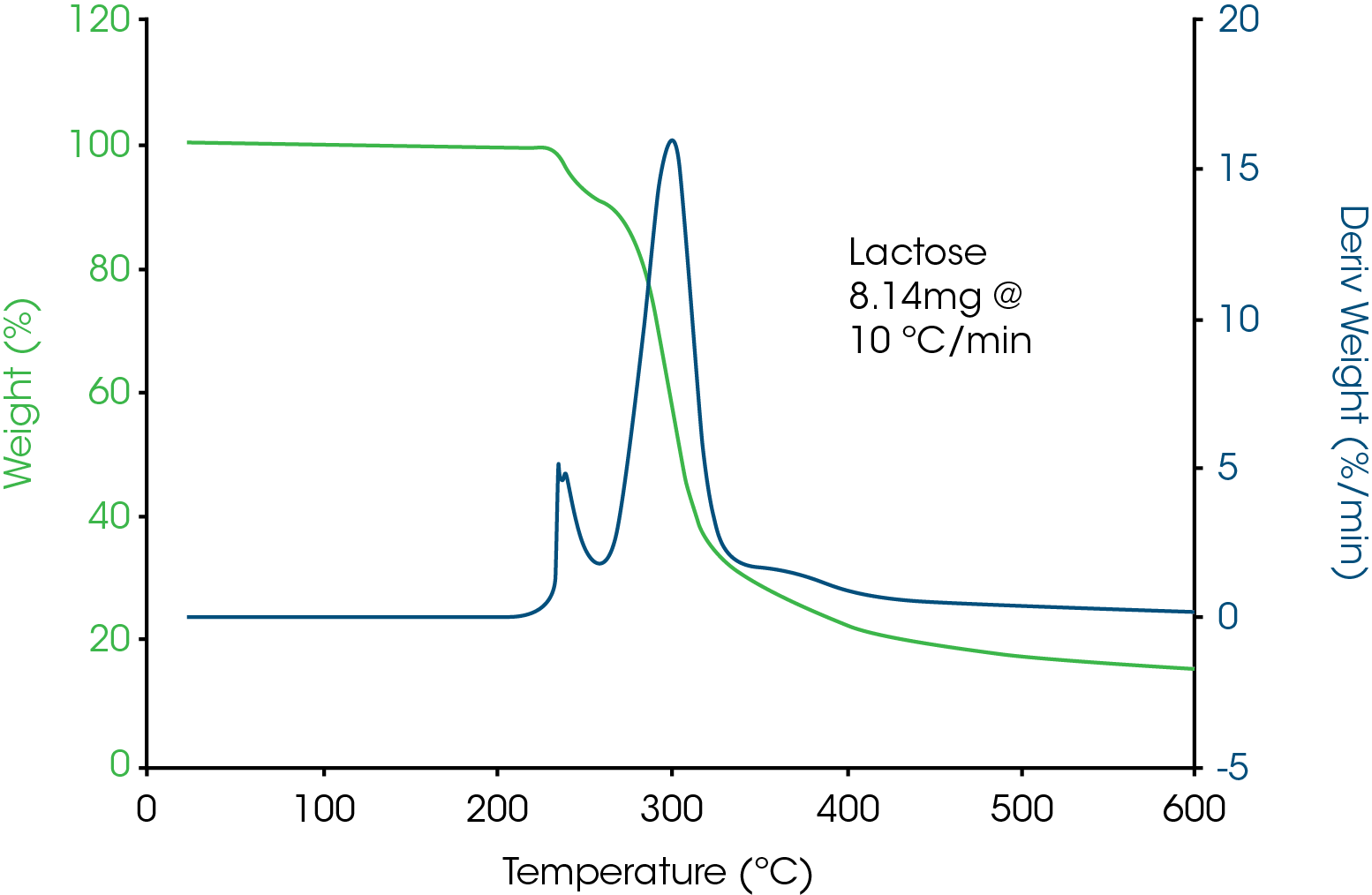
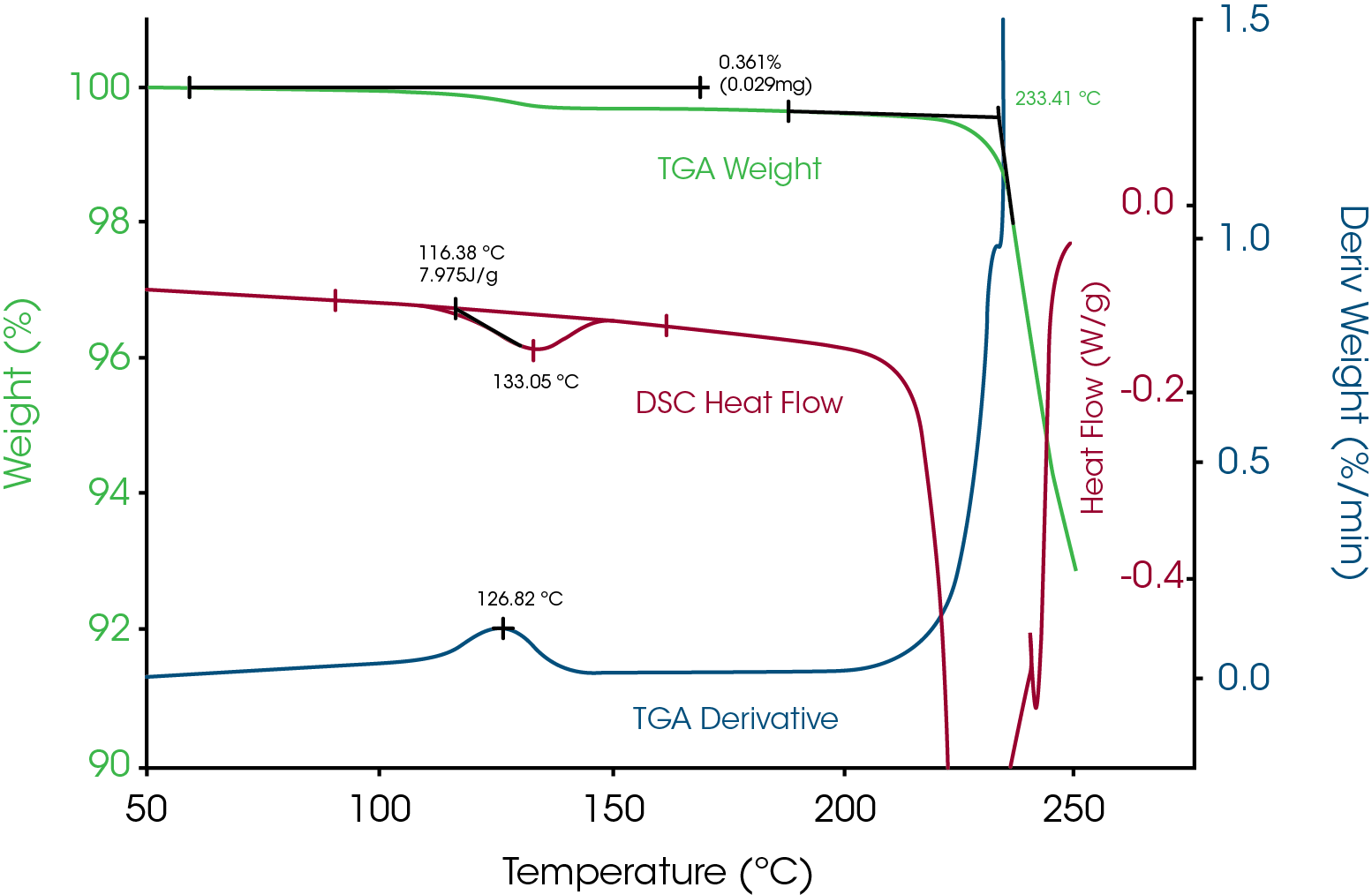
With the knowledge that decomposition does not always cause weight loss, let’s look at a typical TGA analysis on lactose, a common excipient. Figure 4 shows TGA data on lactose heated at 10°C/min from ambient to 600°C. When that file is selected for analysis, the software will plot auto-scale so that all data is shown. The result is that the data is shown at a low sensitivity and minor weight losses may not be detected. It is strongly recommended that TGA data be plotted so that the top 10% weight loss (90-100%) is expanded to look for minor and possibly important weight losses. The benefit of doing this can be seen in Figure 5 where a minor weight loss is detected and most likely due to the presence of hydrated crystals based on the DSC data. In addition, it can be clearly seen that there is little to no free water since there is no weight loss from ambient to 100°C. This is typical for highly crystalline materials which tend to not be hygroscopic. Weight loss above a few tenths of a percent often indicates the presence of amorphous structure which is hygroscopic.
Bulk weight loss of 10% occurs near 250°C in Figure 5 and for that reason should be the upper limit of the DSC experiment in order to not have the hermetic pan burst in the DSC and possibly cause contamination.
Step 2: DSC Analysis at Multiple Heating Rates on Drug and Excipient
The purpose of using multiple heating rates is to determine if the crystalline materials undergo true/thermodynamic melting or apparent melting. All DSC experiments should be done in hermetic pans for this application. If apparent melting is occurring then a chemical process is causing loss of crystalline structure (see TA401 on Apparent Melting). Since all chemical processes are kinetic, loss of crystalline structure will occur at lower temperatures given a longer time (lower heating rate). Loss of crystalline structure at lower temperatures is problematic since the resulting amorphous structure has higher mobility and higher probability of chemical interaction. The use of multiple heating rates also provides the opportunity to investigate reaction rate by using the data to create an Arrhenius plot and to identify the optimum temperature for doing a quasi-isothermal MDSC® experiment so that chemical analysis can be done to identify the reaction mechanism (see Figure 13).
The interaction between acetylsalicylic acid (aspirin) and magnesium stearate is well documented and described in the pharmaceutical website Pharma Treasures. A link to the website and information on this interaction is provided in Figure 6.
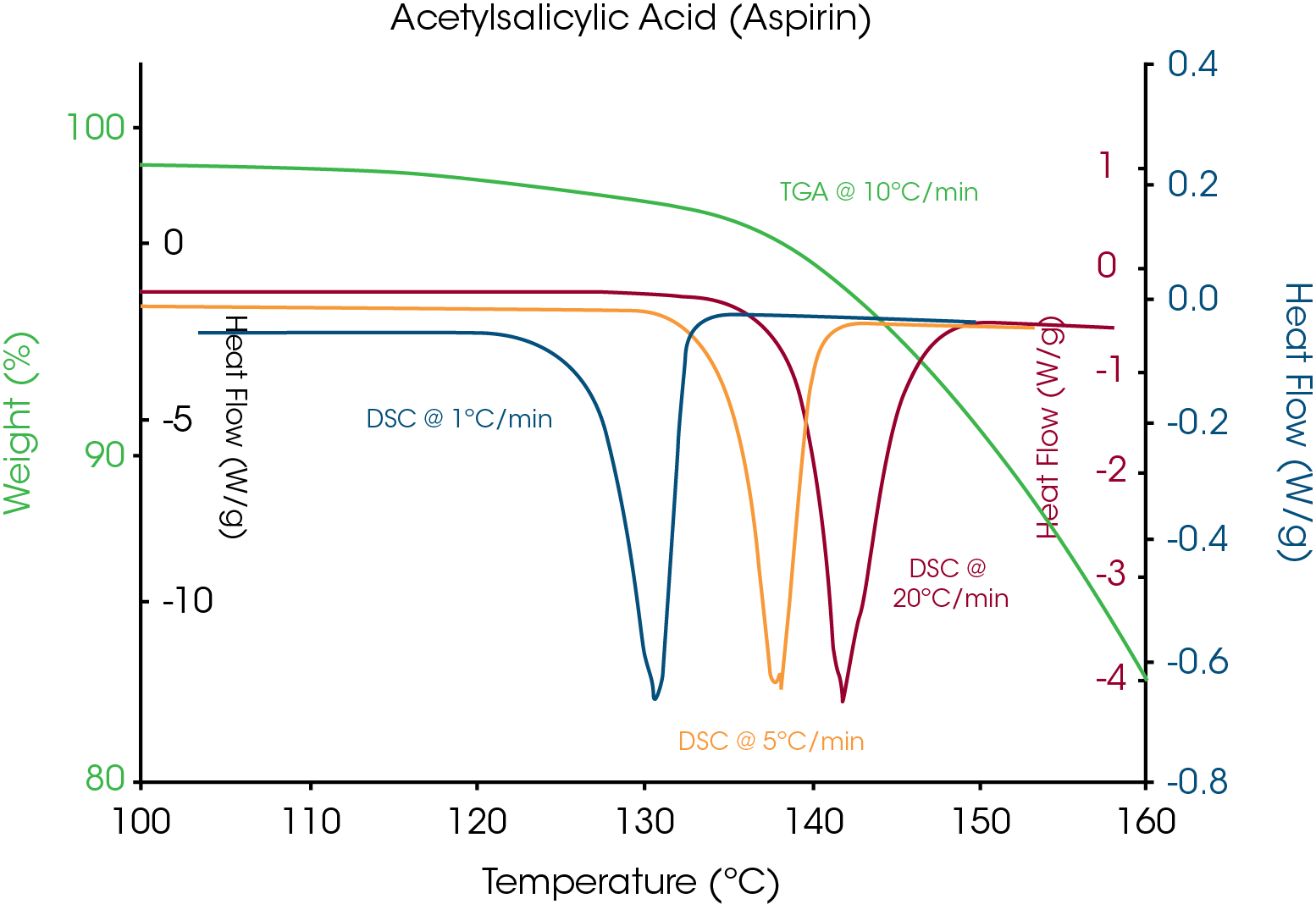
This known drug-excipient incompatibility will be used to illustrate the proposed method. Figure 7 is an overlay of the multiple heating rates for aspirin with the TGA results. The shift in temperature for loss of crystalline structure indicates apparent melting and TGA shows significant weight loss over the same temperature range, indicating decomposition as the cause for loss of crystalline structure.
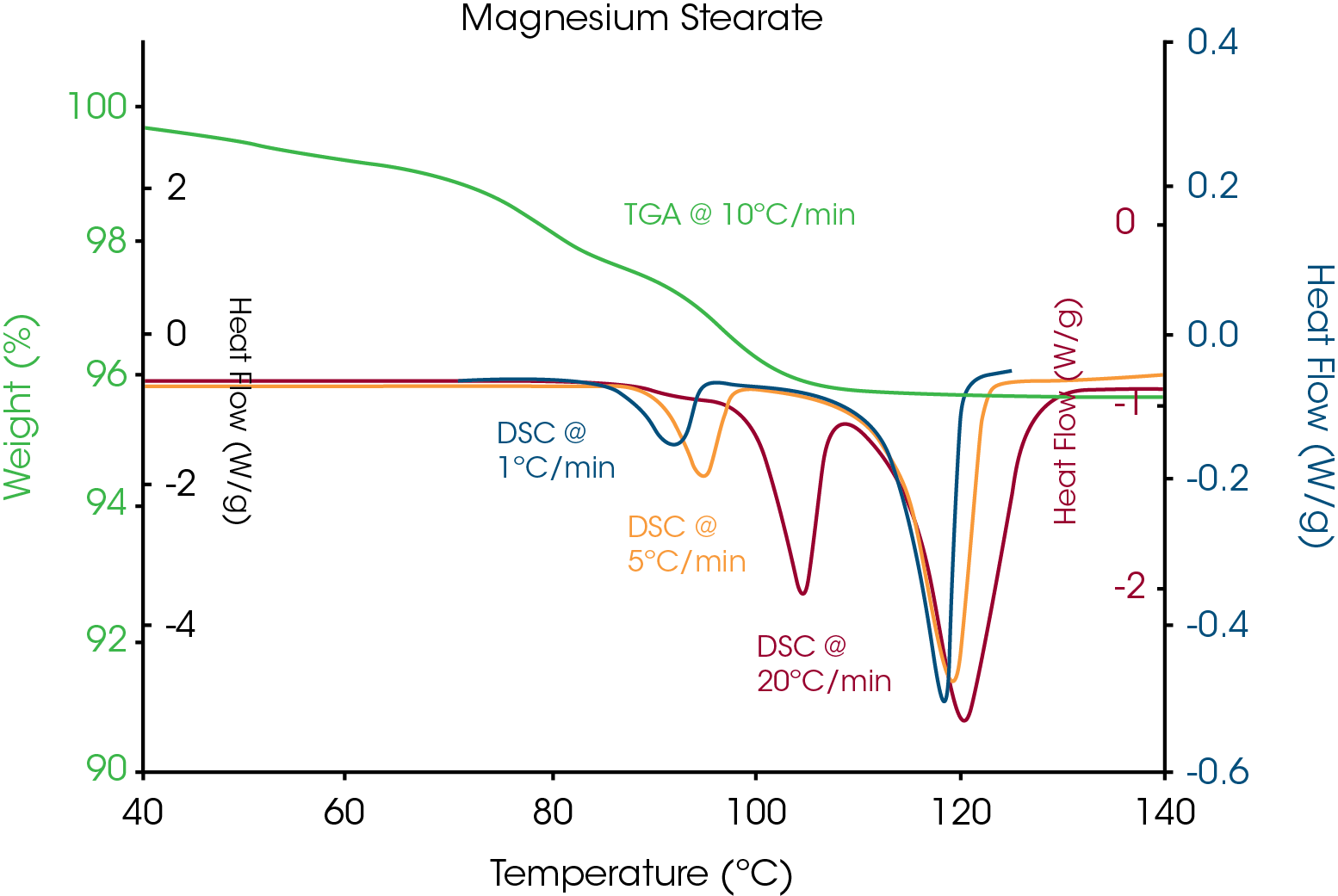
Figure 8 is a similar overlay for the excipient, magnesium stearate. Two endothermic peaks are seen at each heating rate for DSC. The first peak shifts with heating rate and therefore indicates apparent melting. The label on the bottle of the magnesium stearate indicated that it contained both hydrate and anhydrous forms. TGA results show a weight loss in the same temperature range as the first endothermic peaks and this would be expected for dehydration. Since dehydration is a kinetic process, loss of crystallinity due to dehydration would be expected to change with heating rate. The high temperature peaks show no shift with heating rate and there is no weight loss, indicating that the high temperature peak is due to true/thermodynamic melting of the anhydrous form.

Step 3: DSC analysis at 1°C/min on a 50/50 (wt.) mixture of drug and excipient
Once data is obtained for the individual components, it is time to determine if interaction occurs between them in a mixture. The ratio of the two materials is completely arbitrary but a 50/50 mixture by weight is recommended because the sole purpose of the experiment is to detect any interaction. If interaction is detected then other ratios can be investigated. All experiments should be done in hermetic pans.
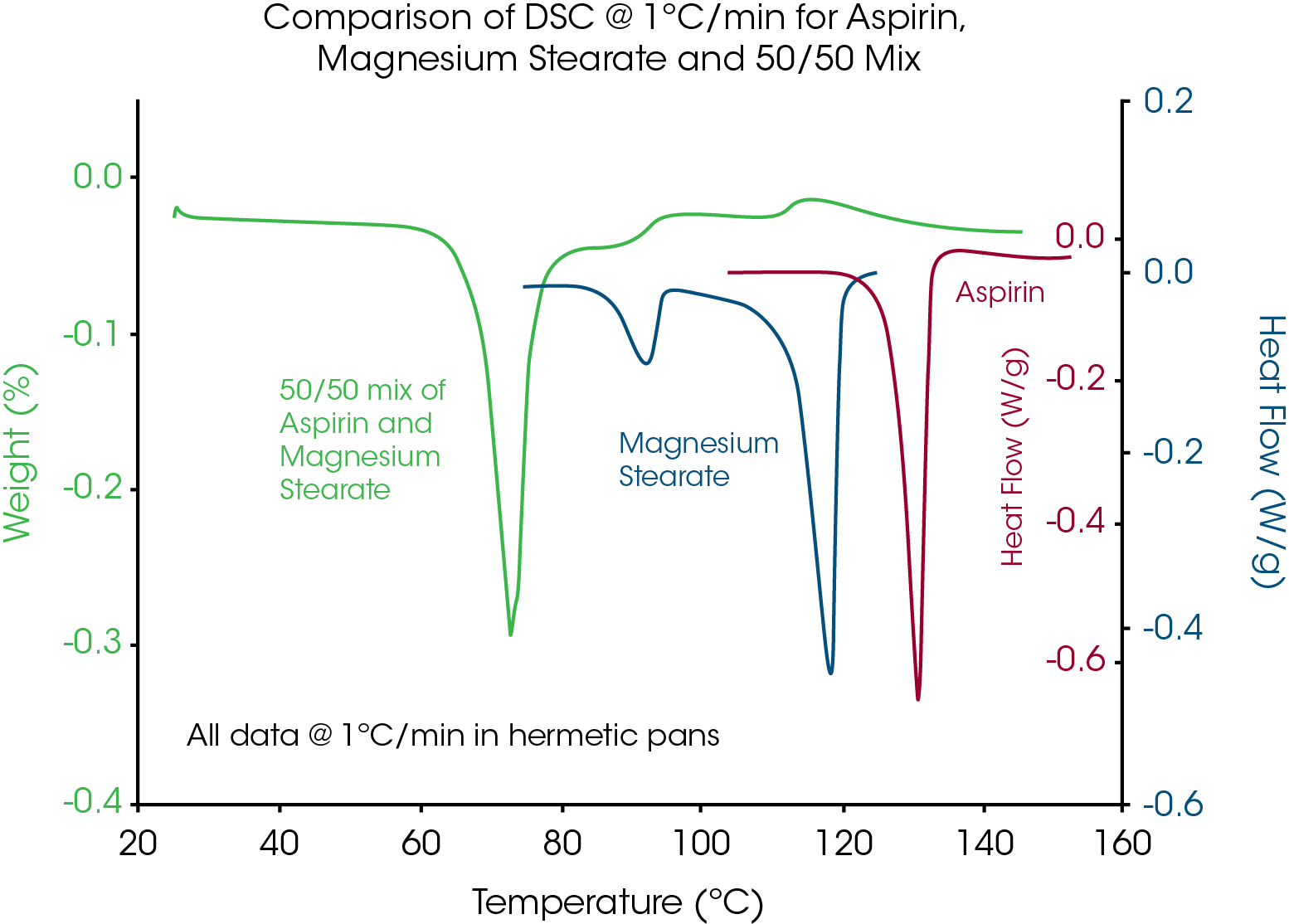
Although it is recommended to make the mixture just prior to the DSC experiment, any interaction, even during mixing or storage, will clearly be seen in the data. The mixture is heated at a slow heating rate (1°C/min) to provide more time for the reaction to occur. If an endothermic peak is seen at a temperature below the melting peaks of the individual components then interaction is indicated as seen in Figure 9 for a 50/50 (wt.) mixture of aspirin and magnesium stearate.
Step 4: Optional Experiments to Investigate Reaction Kinetics or Mechanism
Detection of chemical interaction between an API and potential excipient may eliminate that excipient as a candidate for a final dosage form but there may be interest in understanding the rate of reaction to see how it would affect long-term storage at lower temperatures. To do that requires the determination of reaction kinetics which typically starts with creation of an Arrhenius plot to obtain activation energy and pre-exponential factor. Two ways to obtain kinetic information will be illustrated.
A. Use of Quasi-isothermal MDSC for Analyzing the Rate of Reaction (Kinetics)
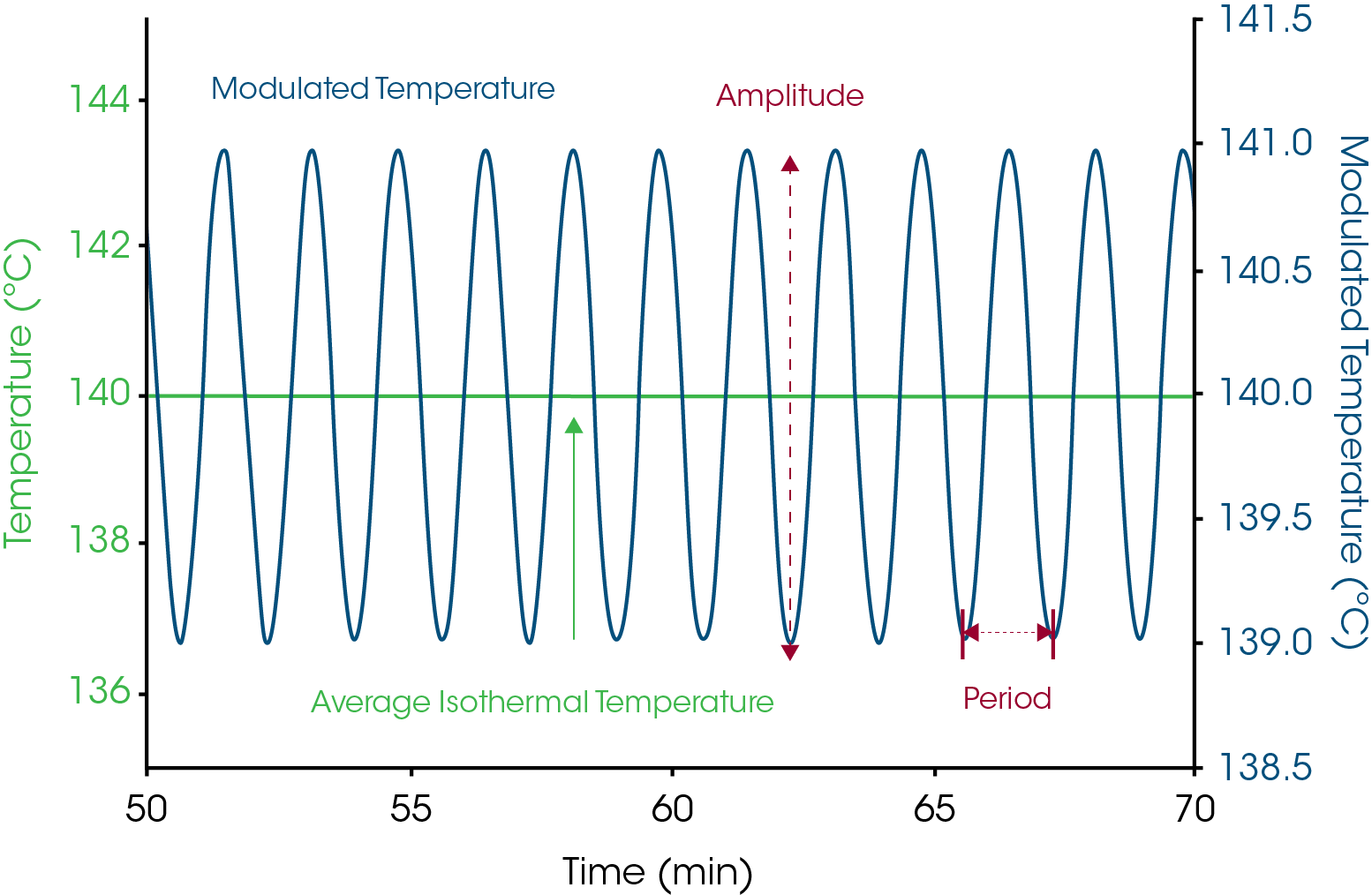
Modulated DSC (MDSC®) is a technique that applies two simultaneous heating rates (linear and sinusoidal) so that heat capacity can be measured at the same time as total heat flow (DSC signal). The sinusoidal heating rate has a temperature modulation amplitude (±°C) and modulation frequency/period (seconds) which can be applied during an isothermal average temperature. The name “quasi-isothermal” simply means that it is not truly isothermal because of the small temperature modulation. An example of such an experiment is seen in Figure 10.
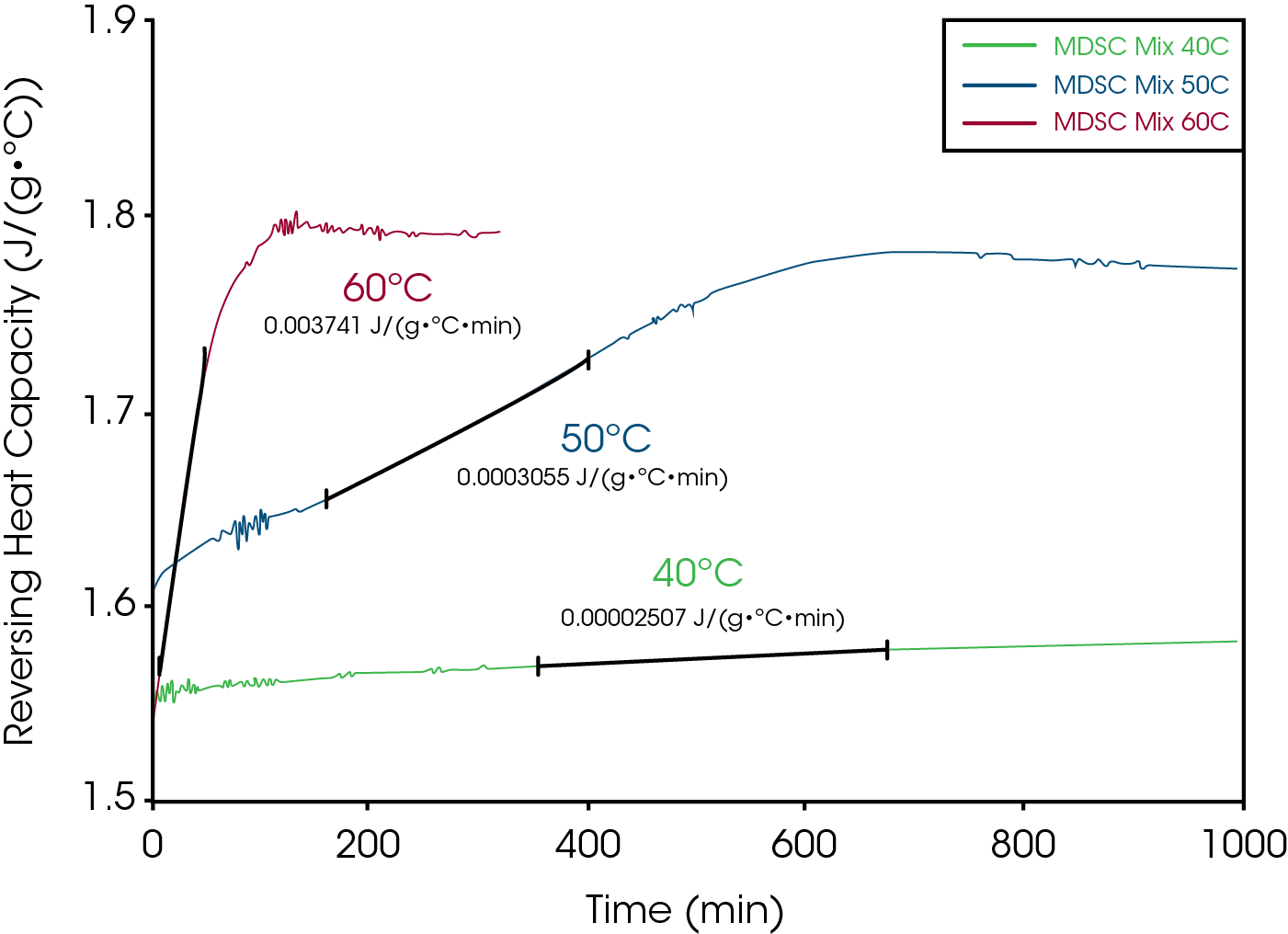
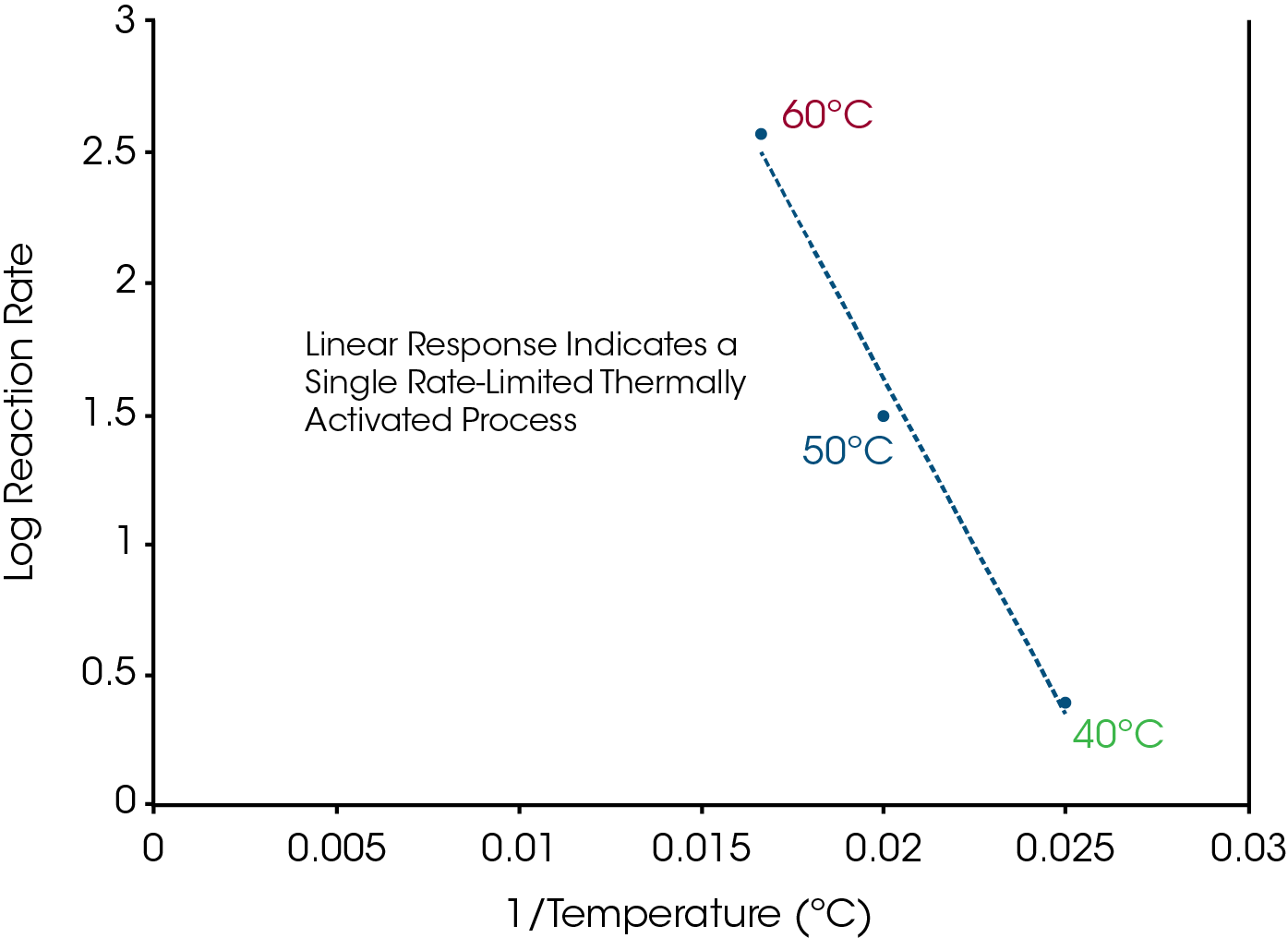
The purpose of measuring heat capacity is to detect conversion of crystalline structure to an amorphous form as a result of the chemical interaction. Amorphous structure typically has a 50% higher heat capacity. Once apparent melting is detected in the 50/50 mixture of drug and excipient, the data can be used to select temperatures for the quasi-isothermal MDSC® experiments. The guideline is to select temperatures in the range between 5 and 20°C below the onset temperature seen in the 50/50 mix experiment at 1°C/min. This is only a guideline since the activation energy varies from one process to another and affects the best isothermal temperatures to use. Based on an onset temperature of approximately 65°C for the 50/50 mix in Figure 10, isothermal temperatures of 40, 50 and 60°C were selected for the quasi-isothermal experiments shown in Figure 114. The data from Figure 11 can be used to create an Arrhenius plot as shown in Figure 12.
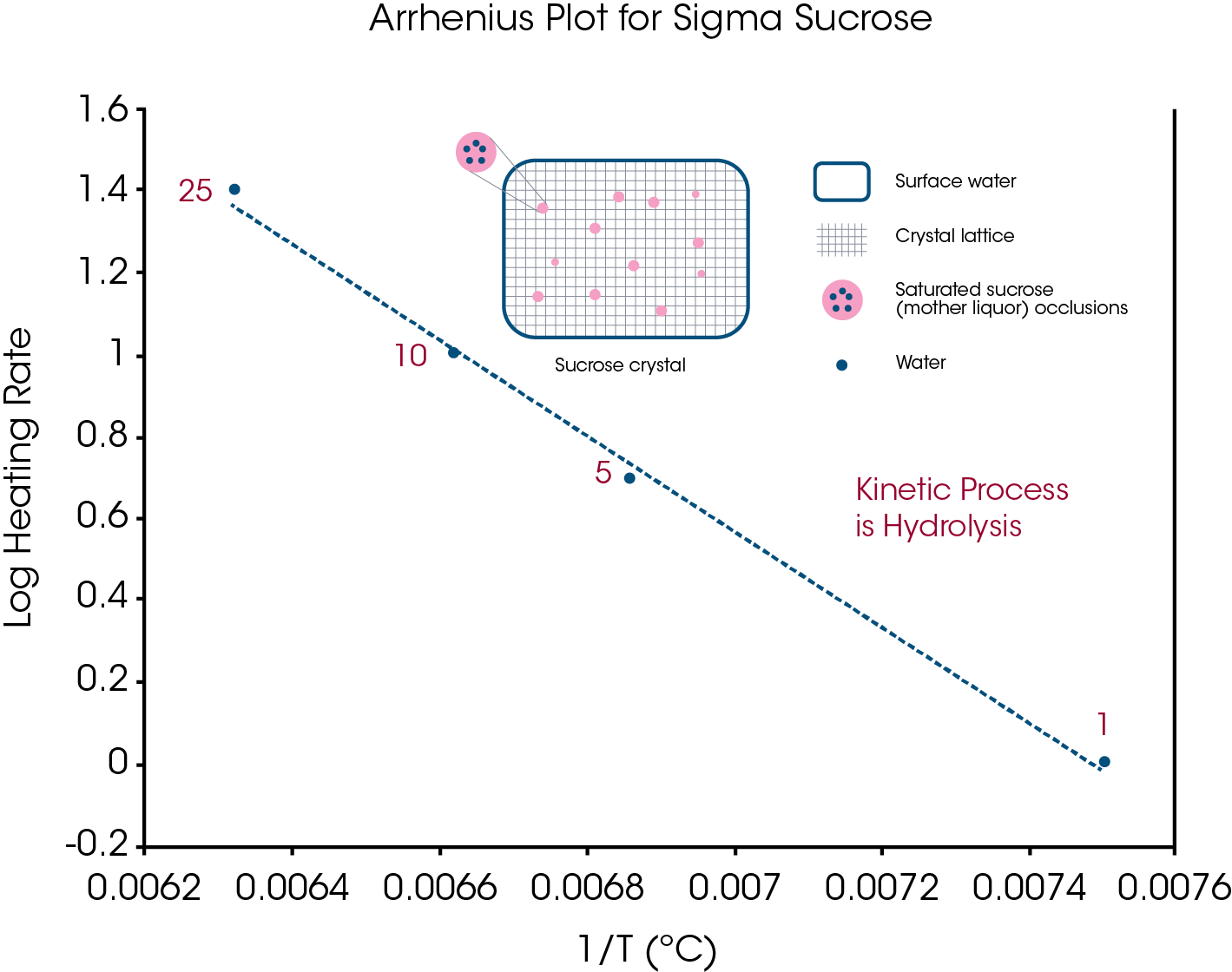
Another way to obtain an Arrhenius plot is to plot log heating rate vs. 1/T for the onset temperatures obtained from multiple heating rates. An example of such a plot is seen in Figure 13 which uses the data from Figure 2 for the apparent melting of sucrose. Except for creating an Arrhenius plot to obtain activation energy and pre-exponential, a detailed discussion of kinetics is outside the scope of this paper.
B. Use of Quasi-isothermal MDSC for Analyzing the Reaction Mechanism (Chemistry)
Once the quasi-isothermal experiments have been completed, the next step is to select a temperature that can be used for preparing a quantity of material at different degrees of conversion that will be used for chemical analysis. Typically, the temperature should result in full loss of crystalline structure in an eight (8) hour period. That way, all samples can be prepared in one day, stored in a refrigerator to stop the reaction, and then analyzed by a chemical analytical technique of choice (HPLC, GC, MS, IR etc.) the next day. An inspection of the quasi-isothermal data for the aspirin-magnesium stearate mix in Figure 11 shows that a temperature of about 52°C should provide the desired result because it took about 600 minutes (10 hours) for complete loss of crystallinity at 50°C. It does not matter if the reaction goes faster or slower at that temperature because the linear response in Figure 11 indicates that a single reaction mechanism is responsible over the temperature range from 40 to 60°C. The goal is to remove samples from the oven at different percent of conversion so that the decomposition mechanism can be followed.
Figure 14 shows chemical analysis by HPLC on the decomposition of sucrose at 120°C 3. The time basis for determining when to remove a new sample from the oven is based on the shape of the Reversing Heat Capacity signal. The shape of the heat capacity curve and rate of creation of the decomposition products (glucose and fructose) may indicate an autocatalytic reaction but that was not seen in the Arrhenius plot.
Conclusions
The proposed thermal analysis method using TGA, DSC and MDSC® is a rapid screening tool for detection of drug-excipient incompatibility. The approach is based on apparent melting which identifies loss of crystalline structure caused by chemical processes such as chemical interaction and decomposition. The current pharmaceutical approach requires months of oven aging and chemical analyses and a relatively large quantity of API and excipient. The proposed method typically takes less than a week, requires less than a gram of material and can provide information on reaction kinetics (rate) and mechanism (chemistry).
References
1. Lee, J.W. et al.; Investigation of the heating rate dependency associated with the loss of crystalline structure in sucrose, glucose, and fructose using a thermal analysis approach (Part I). J. Agric. Food. Chem. 2010. DOI: 10.1021/jf1042344
2. Lee, J.W. et al; Investigation of Thermal Decomposition as the Kinetic Process That Causes the Loss of Crystalline Structure in Sucrose Using a Chemical Analysis Approach (Part II). J. Agric. Food. Chem. 2010. DOI: 10.1021/jf1042344
3. Lu, Y. et al. Impact of Sucrose Crystal Composition and Chemistry on its Thermal Behavior, Journal of Food Engineering 214: 193-208 (2017)
4. Sanchez, Yaritza, TA Instruments; Difference Between “Apparent” Melting and Thermodynamic Melting on Crystalline Materials Using Differential Scanning Calorimetry, presented at 45th NATAS Conference, 2018 in Philadelphia, PA.
Acknowledgement
This note was written by Leonard C. Thomas, DSC Solutions
TA Instruments has been long recognized as an innovator and leader in modulated thermal analysis.
Click here to download the printable version of this application note.

